SPYGLASS IOL STUDY - 2025

Colorado Eye Institute is partnering with SpyGlass Pharma to offer clinical trial opportunities for patients with both Cataracts and Glaucoma.
If you or one of your patients is dealing with both of these conditions and are interested in this clinical study, please reach out via a contact form and our research department will get in contact with you for more information.
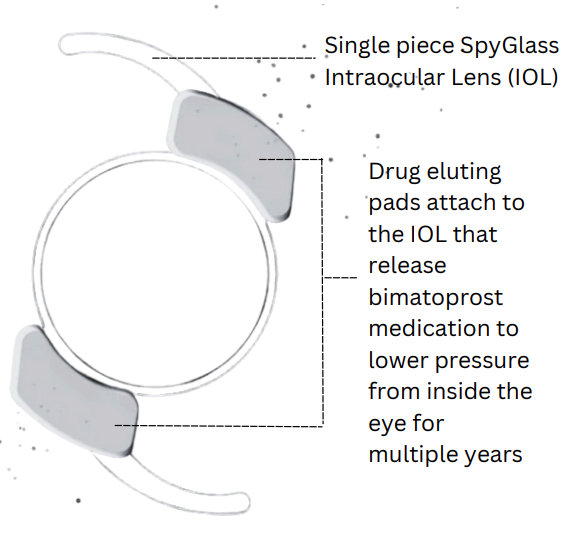
This study is for a new IOL lens for Cataract surgery that provides targeted 3-year delivery of bimatoprost to treat glaucoma in patients undergoing cataract surgery, meaning patients who receive this lens will be able to have glaucoma medication delivered directly from their cataract lens after surgery for up to 3 years after the cataract surgery has been completed.
HOW IT WORKS
The SpyGlass Pharma™ Drug Delivery Platform is implanted with the SpyGlass IOL into the capsular bag via standard cataract surgical technique.
This is a single-piece lens designed for cataract surgery that includes drug-eluting pads releasing bimatoprost to lower intraocular pressure over multiple years. It is made from the same material as standard IOLs and is implanted using typical cataract surgery techniques. The bimatoprost therapy has been FDA-approved for over 20 years. The SpyGlass drug delivery system is currently limited to investigational use under U.S. law. Participants may receive reimbursement for time and travel, as well as eye exams and follow-up care for up to three years. Patients with glaucoma or ocular hypertension should inform their healthcare provider if considering cataract surgery.

